I previously posted regarding a meta-analysis which concluded that the risk of myocarditis after Covid infection is seven times greater than after vaccination. This specific talking point has been used to encourage the injection of a bioweapon into a great many people, and continues to be used to argue its benefit. This particular meta-analysis directly stated vaccine hesitancy as rationale for its necessity.
Given the high rate of vaccine hesitancy due to the fear of vaccine-induced serious adverse events such as cardiac complications, it is critical to characterize the relative risk of vaccine- and infection-induced myocarditis in the general population and determine the effect of sex and age on the risk.
From the 763 records indicated, this meta-analysis included just 22, which is a lot of exclusions.
A commenter accurately pointed out the sloppiness of my summary. He went on to say that I was “too stupid” to see that they gave reasons for the exclusions, which they technically did.
He hopes that I’ve learned my lesson and will read studies more carefully in the future.
Noted.
So today I’m taking a moment to read things a little more carefully. In doing this, I noticed that 14 of the 22 included studies had disclosure statements of interest.
There are a lot of people with a vested interest in myocarditis not being associated with the injections. Turns out, those very same people are often the ones doing the science on myocarditis not being associated with the injections.
Financial conflict of interest can be direct, such as a company funding a drug trial for a drug that they produce.
Or it can be indirect. For example, the authors of a study could own stock in a company that is affected by the outcome of the study they are performing.
This particular meta-analysis was quite large, and included 58,000,000 total study subjects. 53,815,945 of them came from studies that had potentially conflicting interests worthy of disclosure.
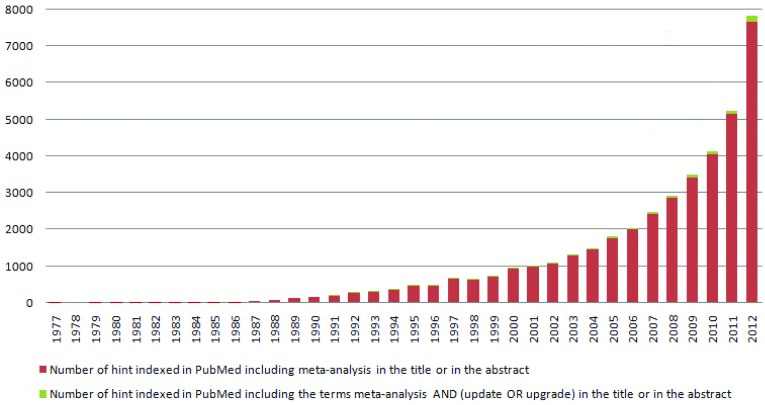
Let’s talk about meta-analyses for a sec. What I have noticed is that the meta-analysis often gets used as a definitive; a slam dunk. A talking point to shut down debate.
Popularity of the meta-analysis has grown exponentially in last 25 years, as shown in the following 2013 graph from PubMed:
Now it is considered the gold standard, as if it’s the final word. However the power of the meta analysis and its aggregate data is only as strong as its component parts.
Meta-analysis (PubMed)
Meta-analysis is a powerful tool to cumulate and summarize the knowledge in a research field through statistical instruments, and to identify the overall measure of a treatment’s effect by combining several individual results [4]. However, it is a controversial tool, because several conditions are critical and even small violations of these can lead to misleading conclusions. In fact, several decisions made when designing and performing a meta-analysis require personal judgment and expertise, thus creating personal biases or expectations that may influence the result [5, 6].
The importance of the quality of the studies used in a meta analysis cannot be overstated.
The quality of bad or compromised science doesn’t get better by mixing it with more science.
Garbage in, garbage out and crooked doesn’t get straighter later.
More from PubMed:
The information gained from quality assessment is fundamental to determine the strength of inferences and to assign grades to recommendations generated within a review. The main problem during the quality assessment process is the inconsistent base for judgment: if the studies were re-examined, the same trained investigator might alter category assignments [6]. The investigator may also be influenced (consciously or unconsciously) by other unstated aspects of the studies, such as the prestige of the journal or the identity of the authors [6].
Conflicts of Interest
Conflicts of interest are considered in individual studies, which is why they are disclosed. It stands to reason that they should be considered in the aggregation of data from multiple studies.
I consulted Cochrane.org, a nonprofit learning resource seeking to increase scientific literacy and minimize bias.
Cochrane Reviews seek to minimize bias. We define bias as a systematic error, or deviation from the truth, in results. Biases can lead to under-estimation or over-estimation of the true intervention effect and can vary in magnitude: some are small (and trivial compared with the observed effect) and some are substantial (so that an apparent finding may be due entirely to bias).
From Cochrane:
7.8.1 Characteristics of conflicts of interest#section-7-8-1
The Institute of Medicine defined conflicts of interest as “a set of circumstances that creates a risk that professional judgment or actions regarding a primary interest will be unduly influenced by a secondary interest” (Lo et al 2009).
Financial conflicts of interest involve both financial interests related to a specific trial (for example, a company funding a trial of a drug produced by the same company) and financial interests related to the authors of a trial report (for example, authors’ ownership of stocks or employment by a drug company).
For drug and device companies and other manufacturers, the financial difference between a negative and positive pivotal trial can be considerable.
For example, the mean stock price of the companies funding 23 positive pivotal oncology trials increased by 14% after disclosure of the results (Rothenstein et al 2011).
In other words, financial conflicts of interests can exist in different ways; both by direct employment and in ownership of stock options. We will see examples of both of these in the disclosures presented.
The mean (average) stock price of a company can be directly affected by the results of a trial or study.
The following graphs show that in 2021, the first year of the Covid-19 injection roll out, the stock price of Pfizer, Moderna, and Novavax, three companies you’ll see well represented in study disclosures, were experiencing all time highs.
The all-time high Pfizer stock closing price was 55.87 on December 16, 2021
The all-time high Moderna stock closing price was 484.47 on August 09, 2021
The all-time high Novavax stock closing price was 319.93 on February 08, 2021
Myocarditis rose to the top of the adverse events associated with the Covid-19 injections, and by 2021 was being reported in the mainstream media. This certainly threatened the financial success that was being experienced by those with a vested interest in these “safe and effective” products.
Sooooo….that was a long winded prep for looking at that meta-analysis that said infection was waaaay more likely to cause myocarditis than the jabs.
With awareness of the potential for conflicts of interest to skew results, let’s go back to our meta-analysis on the risk of myocarditis following injection vs myocarditis due to infection and look at some of the financial disclosures.
Myocarditis in SARS-CoV-2 infection vs. COVID-19 vaccination: A systematic review and meta-analysis
Table 1 from the meta-analysis lists the studies included and their characteristics:
In the following section I have copied the citations directly and hyperlinked the relevant studies.
Following the title are any competing interests, disclosures, or conflicts of interest.
Disclosures are listed in narrative form, or in the case of the New England Journal of Medicine, one is directed to the actual disclosure forms, in which case I took screen shots.
3. Patone M, Mei XW, Handunnetthi L, Dixon S, Zaccardi F, Shankar-Hari M, et al. Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection. Nat Med. (2022) 28:410–22. doi: 10.1038/s41591-021-01630-0
Risks of myocarditis, pericarditis, and cardiac arrhythmias associated with COVID-19 vaccination or SARS-CoV-2 infection
Competing interests
A.S. is a member of the Scottish Government Chief Medical Officer’s COVID-19 Advisory Group, the Scottish Government’s Standing Committee on Pandemics and AstraZeneca’s Thrombotic Thrombocytopenic Advisory Group. All roles are unremunerated. J.H.-C. reports grants from NIHR Biomedical Research Centre, Oxford, John Fell Oxford University Press Research Fund and Cancer Research United Kingdom (CR-UK) grant no. C5255/A18085, through the CR-UK Oxford Centre, and grants from the Oxford Wellcome Institutional Strategic Support Fund (204826/Z/16/Z) and other research councils during the conduct of the study. J.H.-C. is an unpaid director of QResearch, a not-for-profit organization that is a partnership between the University of Oxford and EMIS Health who supplied the QResearch database used for this work. J.H.-C. is a founder and shareholder of ClinRisk Ltd and was its medical director until 31 May 2019. ClinRisk Ltd produces open and closed source software to implement clinical risk algorithms (outside this work) into clinical computer systems. J.H.-C. is chair of the NERVTAG risk stratification subgroup and a member of Scientific Advisory Group for Emergencies COVID-19 groups and the NHS group advising on prioritization of use of monoclonal antibodies in SARS-CoV-2 infection. A.H. is a member of the Joint Committee on Vaccination and Immunisation. K.K. is a member of the Governments Scientific Advisory Group for Emergencies. All other authors declare no competing interests related to this paper.
4. Witberg G, Barda N, Hoss S, Richter I, Wiessman M, Aviv Y, et al. Myocarditis after Covid-19 vaccination in a large health care organization. N Engl J Med. (2021) 385:2132–9. doi: 10.1056/NEJMoa2110737
Myocarditis after Covid-19 Vaccination in a Large Health Care Organization
17. Mevorach D, Anis E, Cedar N, Bromberg M, Haas EJ, Nadir E, et al. Myocarditis after BNT162b2 mRNA vaccine against Covid-19 in Israel. N Engl J Med. (2021) 385:2140–9. doi: 10.1056/NEJMoa2109730
Myocarditis after BNT162b2 mRNA Vaccine against Covid-19 in Israel
18. Barda N, Dagan N, Ben-Shlomo Y, Kepten E, Waxman J, Ohana R., et al. Safety of the BNT162b2 mRNA Covid-19 vaccine in a nationwide setting. N Engl J Med. (2021) 385:1078–90. doi: 10.1056/NEJMoa2110475
Safety of the BNT162b2 mRNA Covid-19 Vaccine in a Nationwide Setting
19. Walter EB, Talaat KR, Sabharwal C, Gurtman A, Lockhart S, Paulsen GC, et al. Evaluation of the BNT162b2 Covid-19 vaccine in children 5 to 11 years of age. N Engl J Med. (2022) 386:35–46. doi: 10.1056/NEJMoa2116298
Evaluation of the BNT162b2 Covid-19 Vaccine in Children 5 to 11 Years of Age
20. El Sahly HM, Baden LR, Essink B, Doblecki-Lewis S, Martin JM, Anderson EJ, et al. Efficacy of the mRNA-1273 SARS-CoV-2 vaccine at completion of blinded phase. N Engl J Med. (2021) 385:1774–85. doi: 10.1056/NEJMoa2113017
Efficacy of the mRNA-1273 SARS-CoV-2 Vaccine at Completion of Blinded Phase
21. Heath PT, Galiza EP, Baxter DN, Boffito M, Browne D, Burns F, et al. Safety and efficacy of NVX-CoV2373 Covid-19 vaccine. N Engl J Med. (2021) 385:1172–83. doi: 10.1056/NEJMoa2107659
Safety and Efficacy of NVX-CoV2373 Covid-19 Vaccine
Funded by Novavax; EudraCT number, 2020-004123-16
22. Ali K, Berman G, Zhou H, Deng W, Faughnan V, Coronado-Voges M, et al. Evaluation of mRNA-1273 SARS-CoV-2 vaccine in adolescents. N Engl J Med. (2021) 385:2241–51. doi: 10.1056/NEJMoa2109522
Evaluation of mRNA-1273 SARS-CoV-2 Vaccine in Adolescents
Funded by Moderna and the Biomedical Advanced Research and Development Authority; Teen COVE ClinicalTrials.gov number, NCT04649151
Observer-blind
23. Dunkle LM, Kotloff KL, Gay CL, Áñez G, Adelglass JM, Barrat Hernández AQ, et al. Efficacy and safety of NVX-CoV2373 in adults in the United States and Mexico. N Engl J Med. (2021) 386:531–43. doi: 10.1056/NEJMoa2116185
Efficacy and Safety of NVX-CoV2373 in Adults in the United States and Mexico
24. Diaz GA, Parsons GT, Gering SK, Meier AR, Hutchinson IV, Robicsek A. Myocarditis and pericarditis after vaccination for COVID-19. Jama. (2021) 326:1210–2.
Myocarditis and Pericarditis After Vaccination for COVID-19
26. Husby A, Hansen JV, Fosbøl E, Thiesson EM, Madsen M, Thomsen RW, et al. SARS-CoV-2 vaccination and myocarditis or myopericarditis: population based cohort study. BMJ. (2021) 375:e068665. doi: 10.1136/bmj-2021-068665
SARS-CoV-2 vaccination and myocarditis or myopericarditis: population based cohort study
Ethical approval
The study was conducted using administrative register data. According to Danish law, ethical approval is not required for such research.
Competing Interests
All authors have completed the ICMJE uniform disclosure form at www.icmje.org/disclosure-of-interest/ and declare: support from the Lundbeck Foundation for the submitted work; MA declares that he has previously participated in research projects funded by Pfizer, Janssen, AstraZeneca, H Lundbeck and Mertz, and Novartis with grants received by Karolinska Institutet (no personal fees); MA has personally received fees for teaching from Atrium, the Danish Association of the Pharmaceutical Industry; the Pharmacovigilance Research Centre is supported by a grant from the Novo Nordisk Foundation (NNF15SA0018404) to the University of Copenhagen.
29. Murk W, Gierada M, Fralick M, Weckstein A, Klesh R, Rassen JA. Diagnosis-wide analysis of COVID-19 complications: an exposure-crossover study. Cmaj. (2021) 193:E10–8. doi: 10.1503/cmaj.201686
Diagnosis-wide analysis of COVID-19 complications: an exposure-crossover study
Funding: This study was funded by Aetion, Inc., a manufacturer of software for the analysis of real-world data. Michael Fralick received the Banting and Best Canadian Institute of Health Research Grant.
34. Buckley BJ, Harrison SL, Fazio-Eynullayeva E, Underhill P, Lane DA, Lip GY. Prevalence and clinical outcomes of myocarditis and pericarditis in 718,365 COVID-19 patients. Eur J Clin Invest. (2021) 51:e13679.
Prevalence and clinical outcomes of myocarditis and pericarditis in 718,365 COVID-19 patients
CONFLICTS OF INTEREST
Benjamin JR Buckley has received funding from Bristol-Myers Squibb (BMS)/Pfizer. Stephanie L Harrison has received funding from BMS. Elnara Fazio-Eynullayeva and Paula Underhill are employees of TriNetX LLC. Deirdre A Lane has received investigator-initiated educational grants from BMS, has been a speaker for Boehringer Ingeheim and BMS/Pfizer and has consulted for BMS, Boehringer Ingelheim and Daiichi-Sankyo. Gregory YH Lip: consultant for Bayer/Janssen, BMS/Pfizer, Medtronic, Boehringer Ingelheim, Novartis, Verseon and Daiichi-Sankyo and speaker for Bayer, BMS/Pfizer, Medtronic, Boehringer Ingelheim and Daiichi-Sankyo. No fees are directly received personally.
38. Bozkurt B, Kamat I, Hotez PJ. Myocarditis with COVID-19 mRNA vaccines. Circulation. (2021) 144:471–84. doi: 10.1161/CIRCULATIONAHA.121.056135
Myocarditis With COVID-19 mRNA Vaccines
Disclosures Dr Bozkurt: Consultation for Bayer and scPharmaceuticals, Clinical Events Committee for Guide-HF Trial Abbott Pharmaceuticals, and Data Safety Monitoring Board for Anthem Trial by Liva Nova Pharmaceuticals. Dr Hotez: Inventor on a COVID-19 vaccine technology owned by Baylor College of Medicine that was licensed nonexclusively to vaccine companies in India (Biological E) and elsewhere. Dr Kamat reports no conflicts.
A last word from Cochrane about bias and conflicts of interest:
Both the risk of bias in included studies and risk of bias due to missing results may be influenced by conflicts of interest of study investigators or funders. For example, investigators with a financial interest in showing that a particular drug works may exclude participants who did not respond favourably to the drug from the analysis, or fail to report unfavourable results of the drug in a manuscript.
The volume of disclosures and conflicts of interest riddled throughout the studies included in this meta-analysis seems precisely the bias for which Cochrane warns. Although according to Table 1, the risk of bias with each study is low, with the exception of just two, and according to the authors, “the risk of bias was evaluated at the outcome level using the Cochrane Collaboration tool for RCTs and Newcastle-Ottawa Scale for observational studies (9, 10).”
So, there you have it. I guess the risk for bias is low.
You get the science you pay for.
https://ko-fi.com/conspiracysarah
*apologies to
and for my sloppy first round*






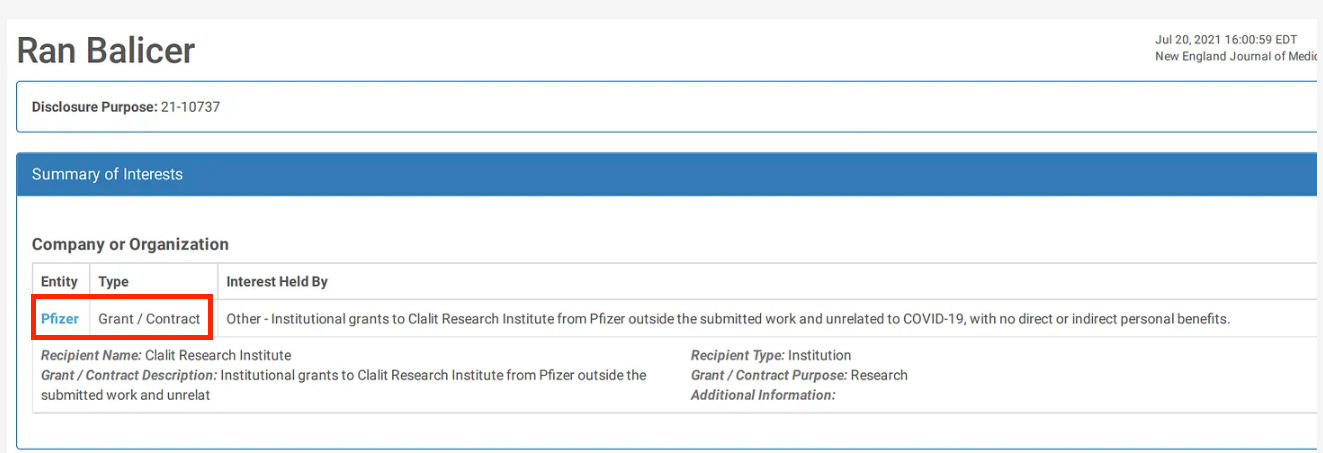
















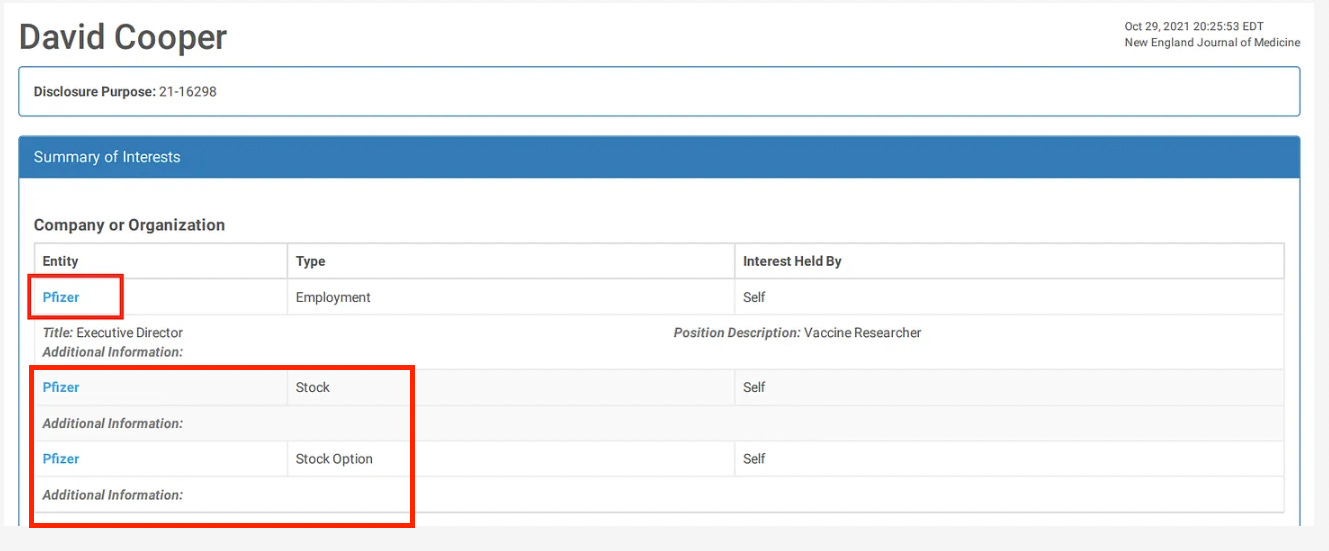

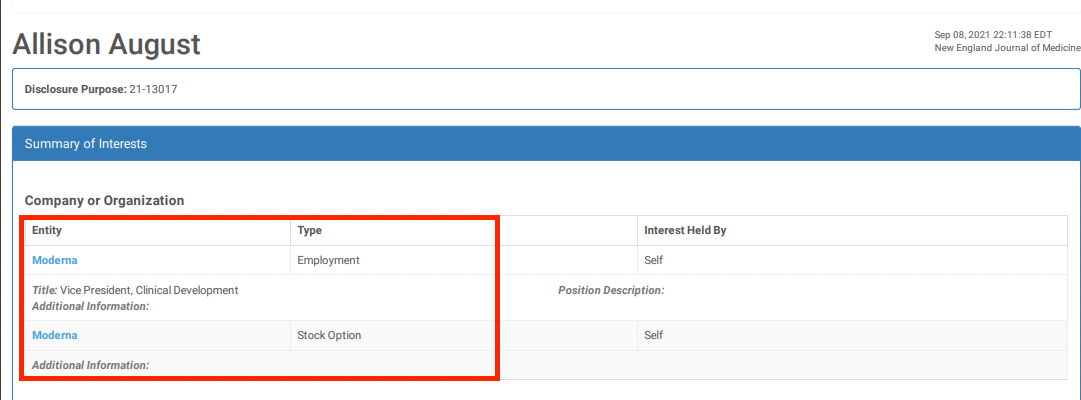








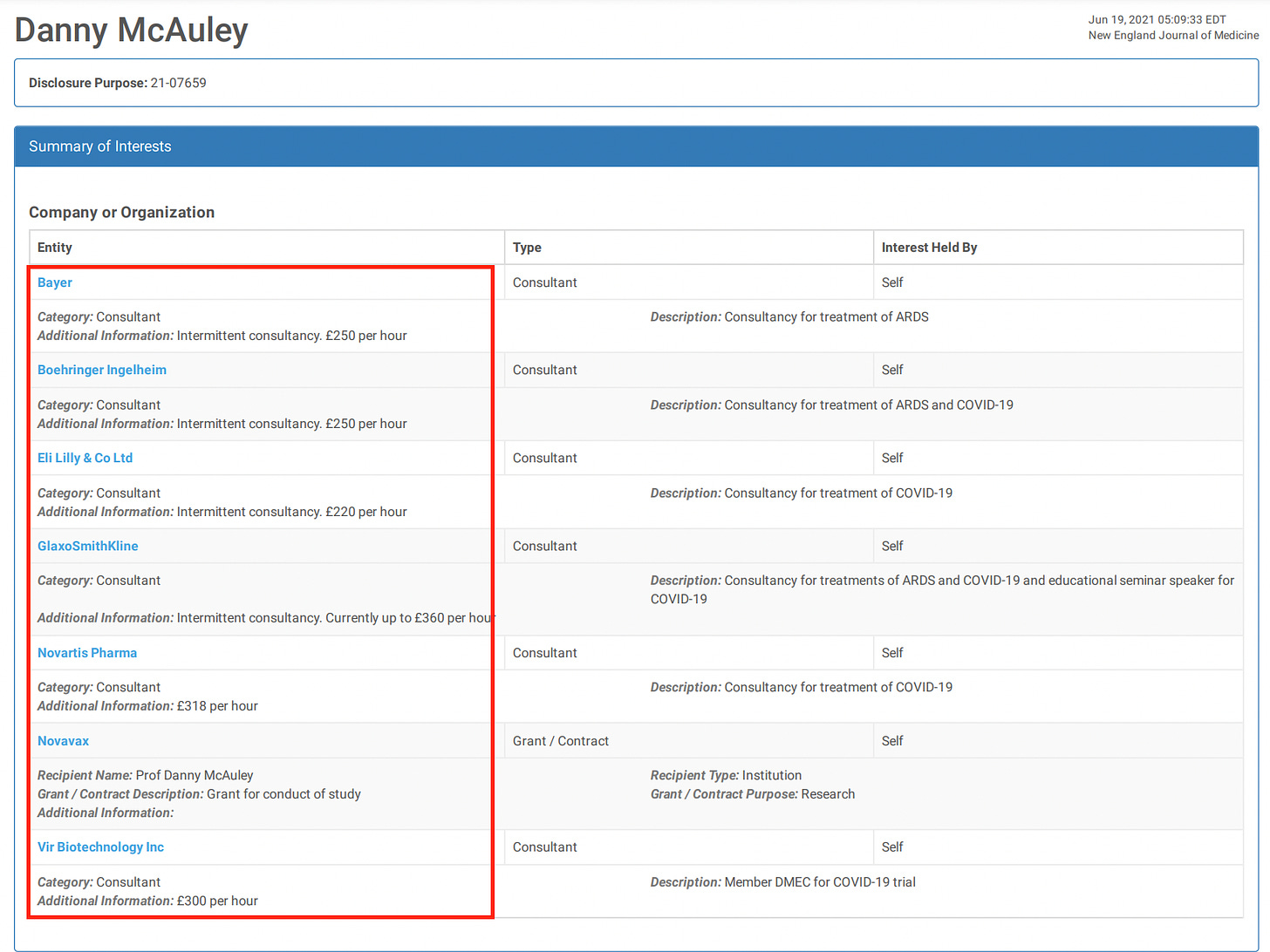











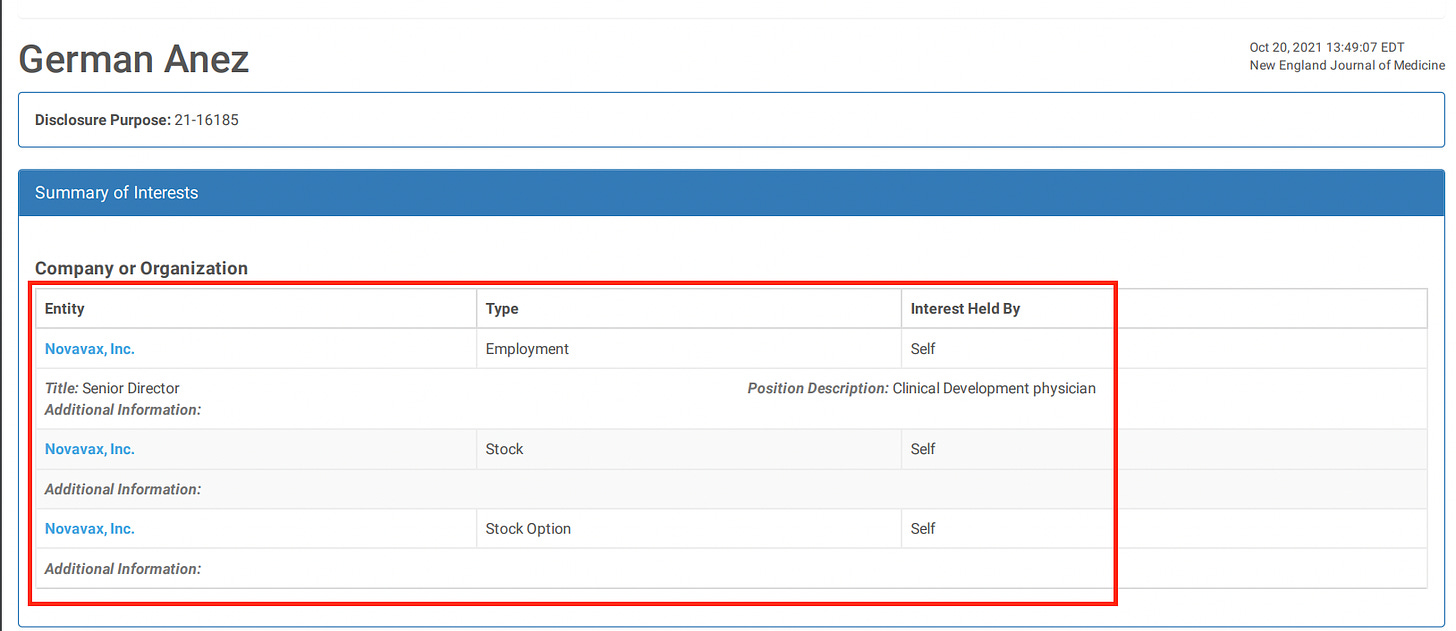





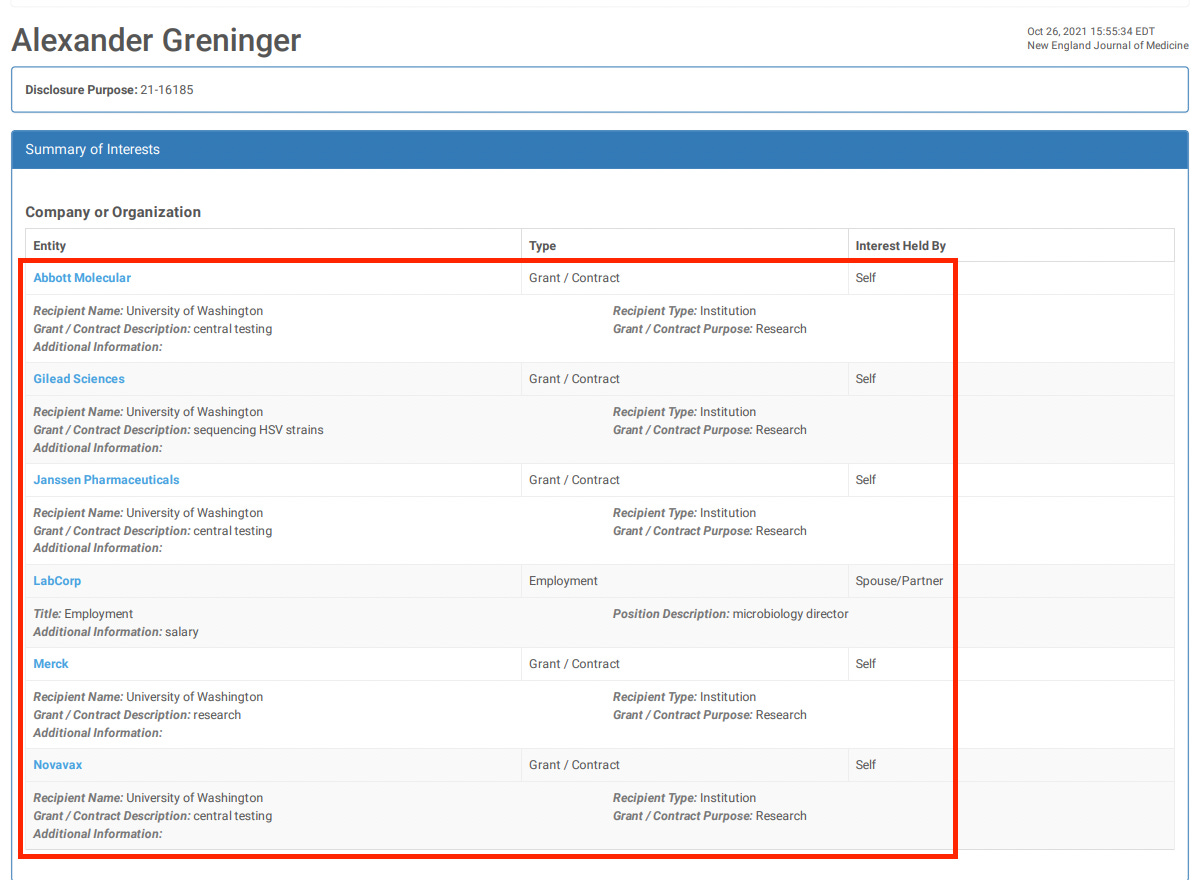








When you get a research grant from big pharma, you also get instructions on what the results had better show.
Absolutely Fabulous Research Sarah !
Endotoxin Induced Myocarditis (EIM) known in fine epigenetic detail since 2003.
https://geoffpain.substack.com/p/pfizer-process-2-endotoxin-myocarditis
Those selling treatments for "Spikeopathy" who fail to mention EIM have a $Millions Conflict of Interest.
I have called that Scientific Fraud by Omission.