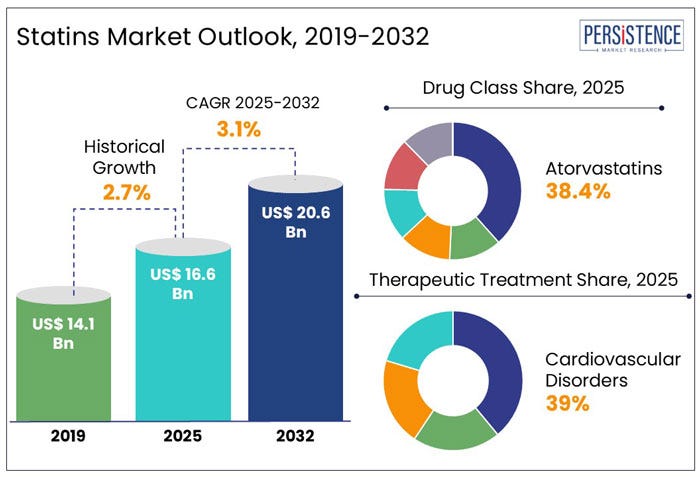
The Cholesterol Industrial Complex
New guidelines: MORE TESTING. EARLIER AND ALWAYS. MORE STATINS.
A client sent this New York Times (🙄) article to me Friday, titled Get Your Cholesterol Even Lower and Start Younger, New Guideline Says.
((((sigh))))
The cholesterol racket apparently knows no bounds and simply will not stop until we have everyone, including children, on a cholesterol lowering drug. The enormity of this ridiculousness cannot be overstated. But the beat goes on. If you would like to know more about this topic I encourage you to read the work of Malcolm Kendrick here and Anthony Colpo (here). Stephan Thomas has a good synopsis of Kendrick’s work here.
Here we go…
March 13, 2026 Updated 3:11 p.m. ET
People at risk of heart disease should start trying to lower their cholesterol as early as age 30 and continue into old age. They should also be getting their cholesterol levels lower than many doctors had previously recommended.
That is the new guidance released Friday by the American Heart Association, the American College of Cardiology and nine other professional organizations.
Before reading any further than the first sentences, I clicked on the “new guidance” hyperlink , which brought me here:
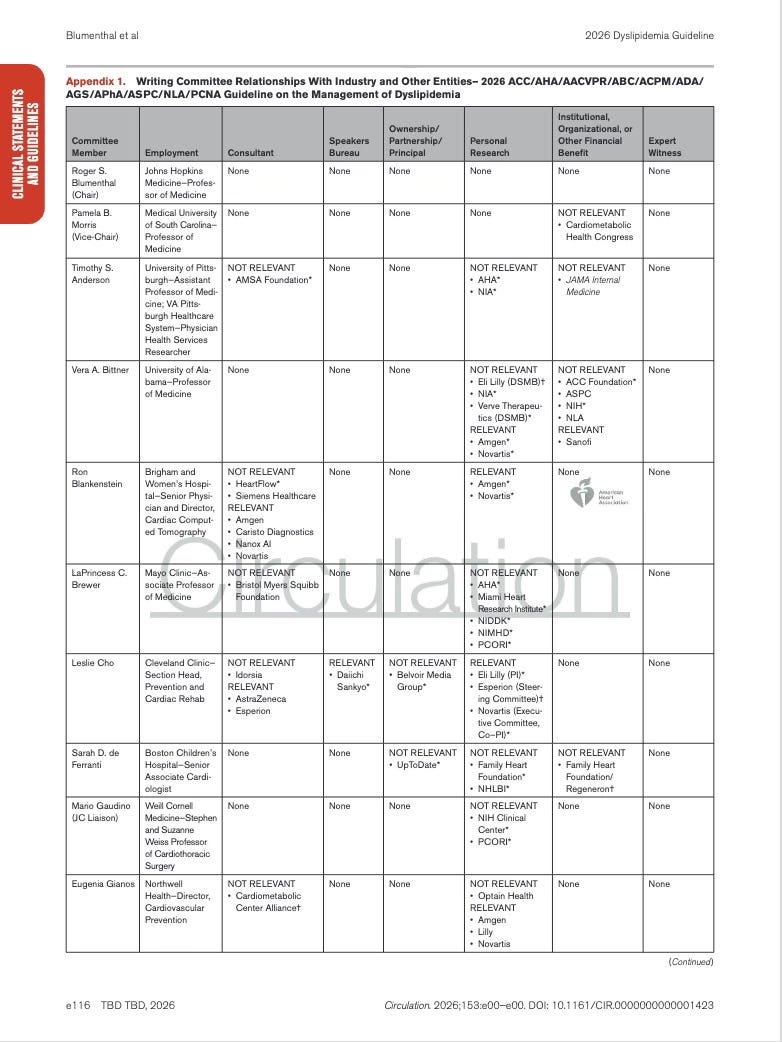
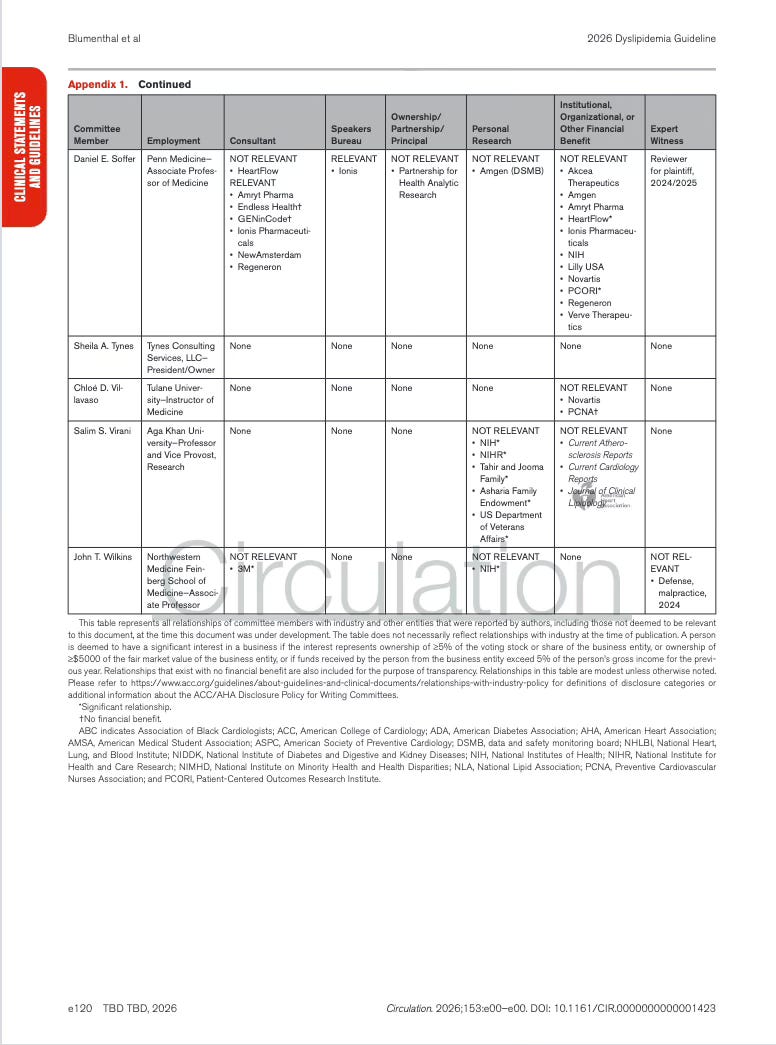
And before reading the new guidelines, I clicked the “Author Info & Affiliations” tab found after the committee member names.
This landed at Appendix 1, which is conveniently buried deep within the document.
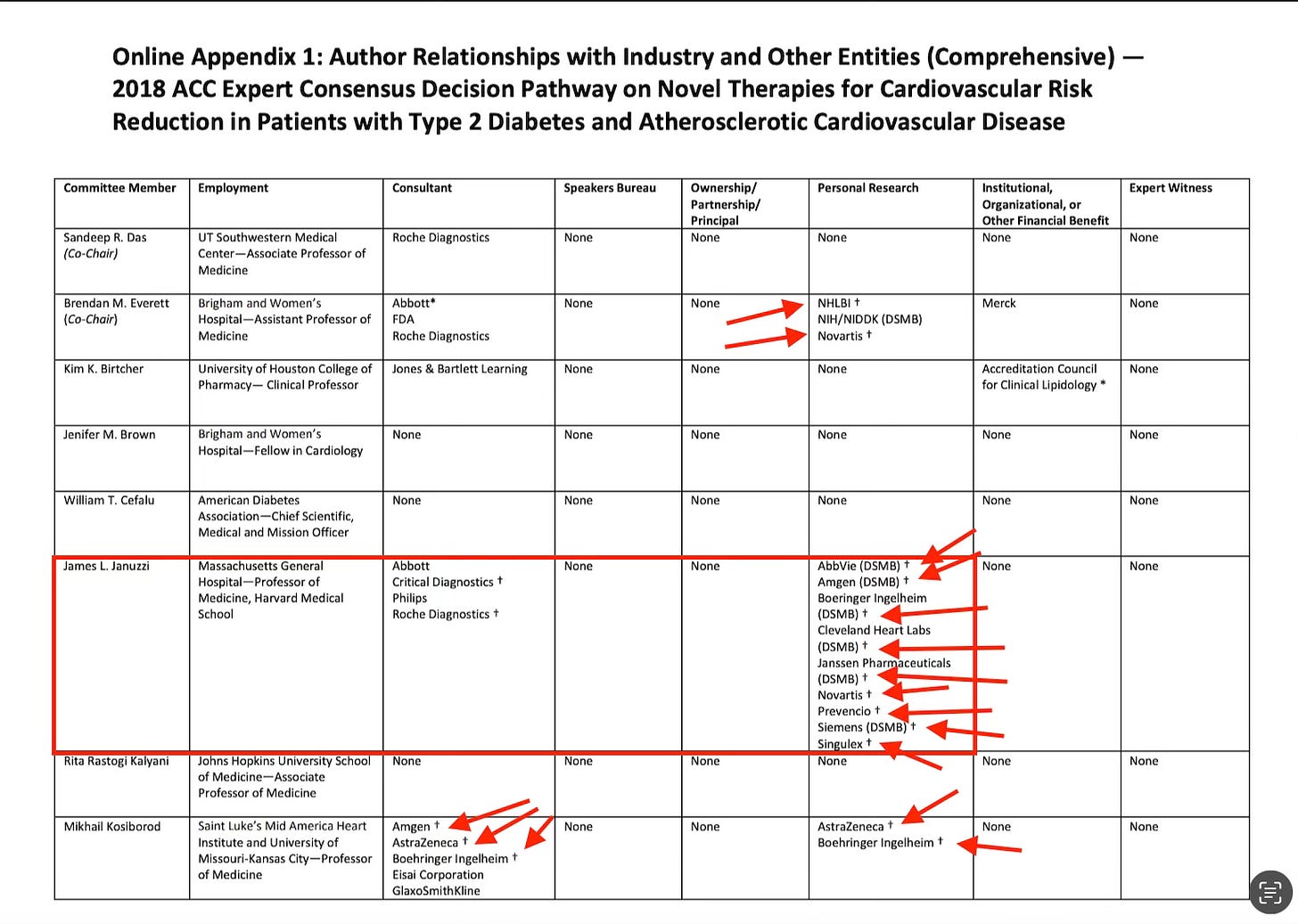
The table lists each member of the guideline writing committee, and documents whether they had any financial or professional relationships with healthcare companies or organizations (yep, those companies whose products are affected by the guideline recommendations.).
Spoiler alert…there were relations.
Note👇
* = Significant relationship
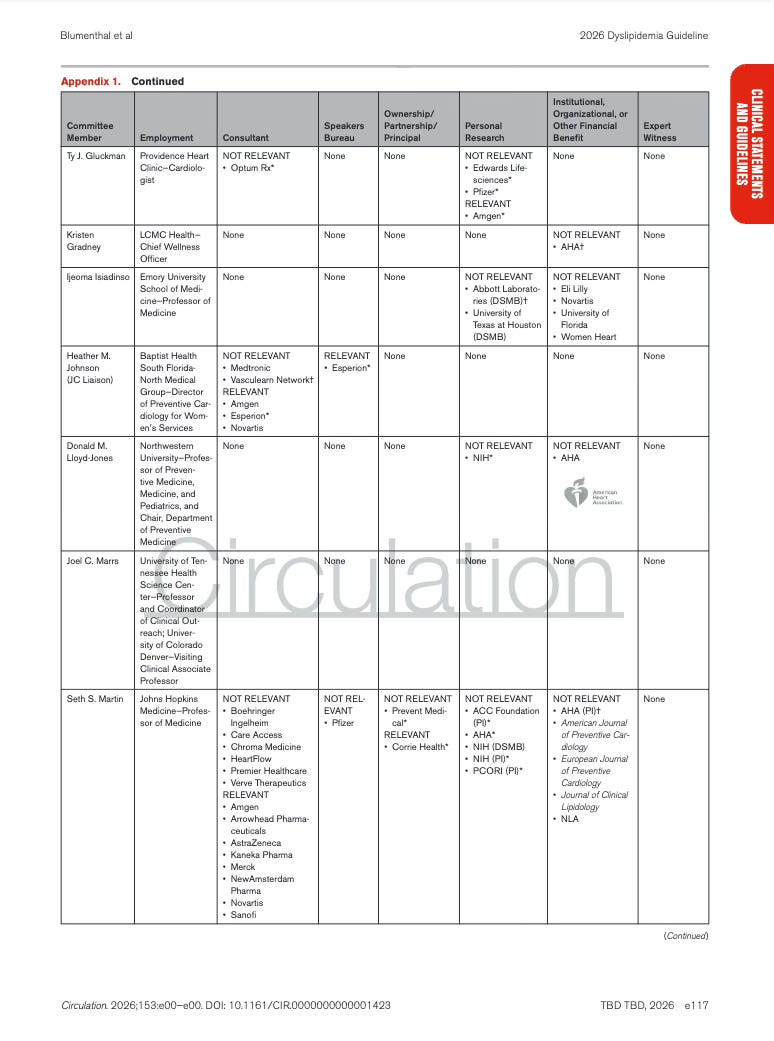
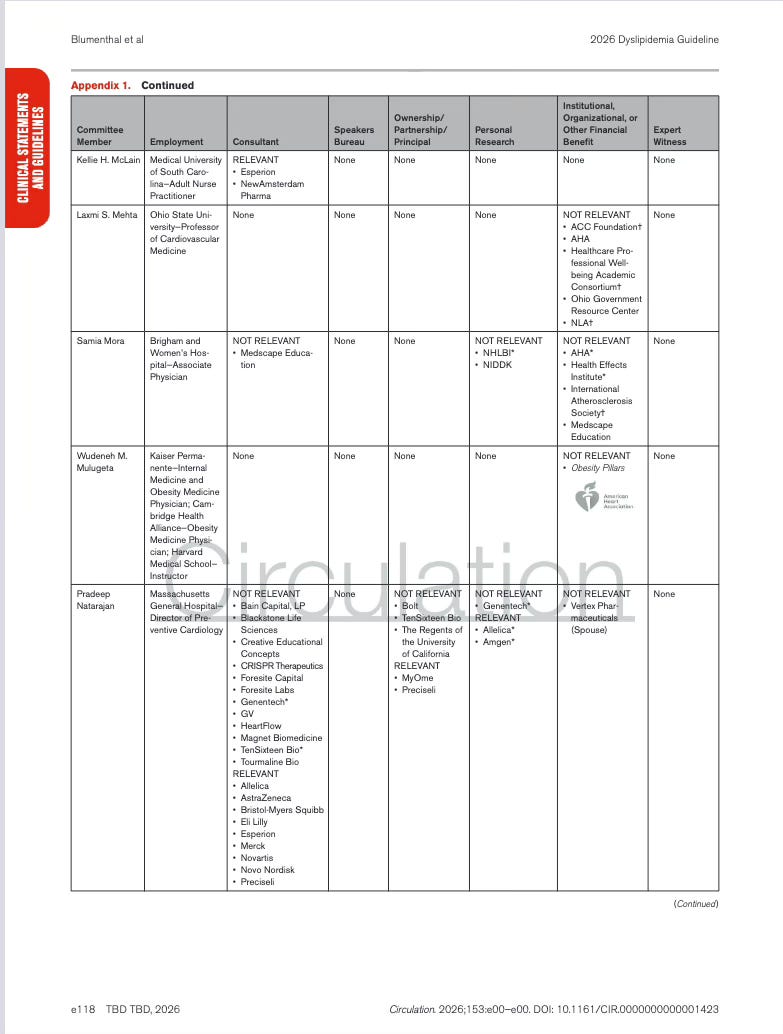
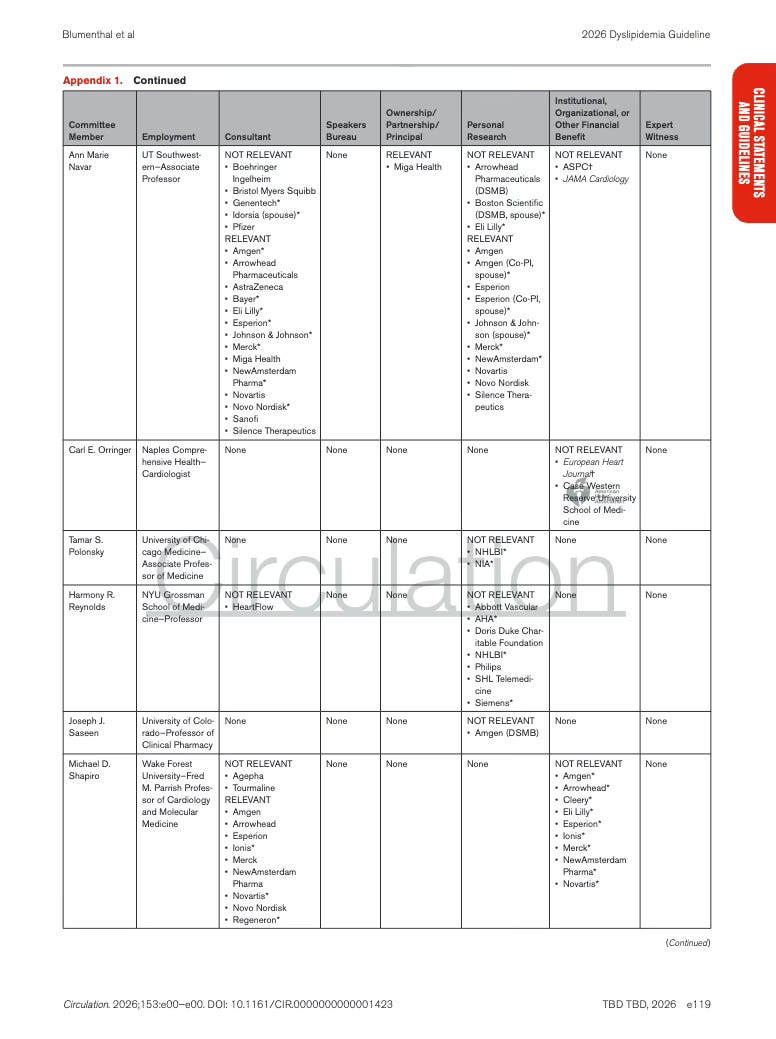
Appendix 1. Writing Committee Relationships With Industry and Other Entities
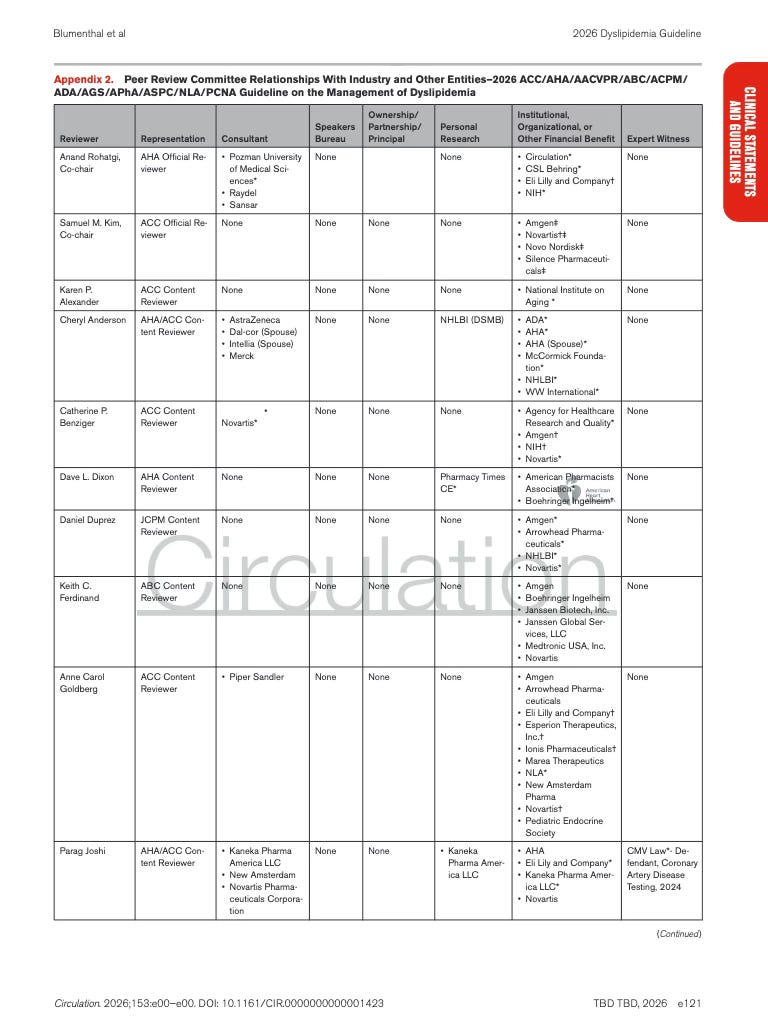
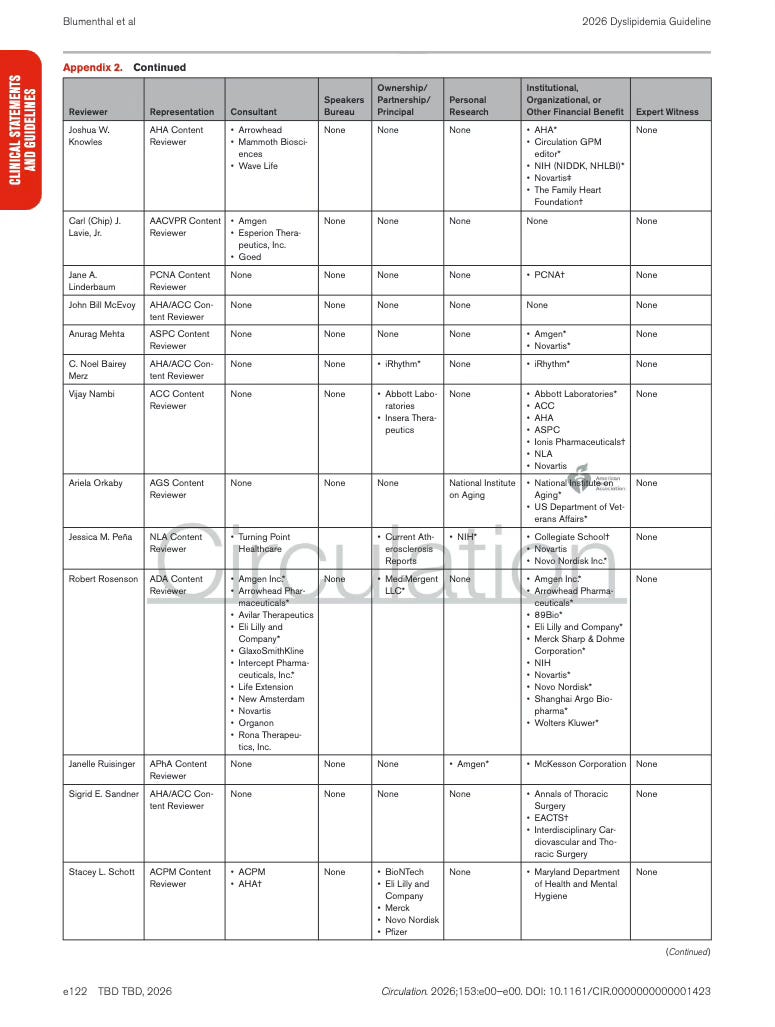
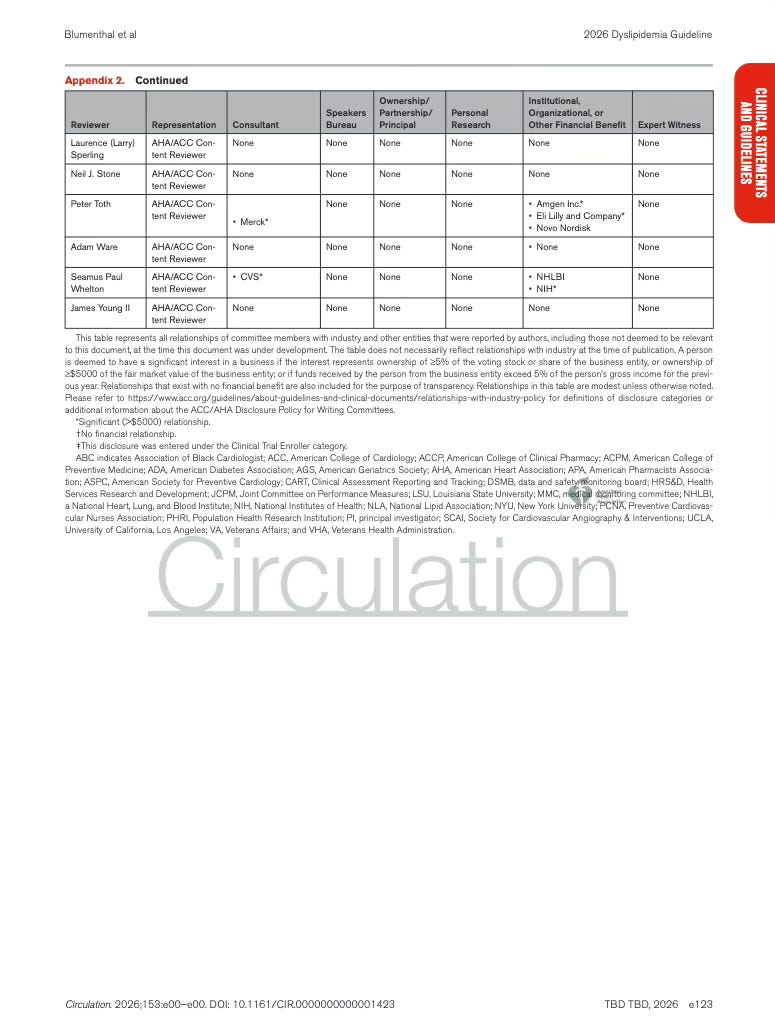
Next up is Appendix 2. This section lists the physicians and organizations that reviewed the guideline before publication and discloses any financial relationships they reported with healthcare companies.
The companies appearing in these disclosures are typically the same pharmaceutical manufacturers whose drugs are discussed in the guideline … particularly those producing cholesterol lowering therapies like statins or lipid lowering agents.
Appendix 2. Peer Review Committee Relationships With Industry and Other Entities
To be crystal clear… the documents above list the financial relationships (conflicts of interest) of both the individuals who wrote the guidelines and the reviewers who evaluated them. These disclosures identify any consulting work, research funding, advisory roles, or other professional ties those individuals have had with pharmaceutical companies whose drugs or products are addressed within the guidelines themselves.
From my estimation, and as I have documented many times previously, there is a very clear pattern that has emerged across the medical-pharma-industrial complex that looks like this:
And this familiar pattern was certainly evident the last time the ACC/AHA cholesterol guidelines were updated in 2018. The industry response following those guidelines was almost immediate. This is when add-on cholesterol drugs (ezetimibe and PCSK9 inhibitors) became all the rage. The authors bellyached within the document about the price of PCSK9 inhibitors, which at the time were considered pricey. They said “JUMP PHARMA”, and within months, the manufacturers said “HOW HIGH???” Amgen cut the list price of Repatha by about 60%, and Sanofi/Regeneron followed with Praluent. Once the price point decreased, the market expanded, and new drugs came down the pipeline … prescribing increased, and combination lipid therapy became the norm.
The conflict of interest picture was not as overt as we sometimes see, but it wasn’t exactly covert either. The 2018 guideline’s writing committee reported no relevant financial relationships during the development window, including the chair. But the peer-review appendix sings a different song. Several reviewers disclosed consulting, research funding, or advisory relationships with companies that manufacture cholesterol drugs… you know, like Amgen, Sanofi, Regeneron, Merck, Pfizer, and Esperion. More importantly, the evidence base driving the guidelines came heavily from industry funded trials such as FOURIER and ODYSSEY OUTCOMES. In other words, the guidelines were technically written without direct pharmaceutical industry funding … but The Science™ propping those guidelines up was absolutely industry funded.
For example:
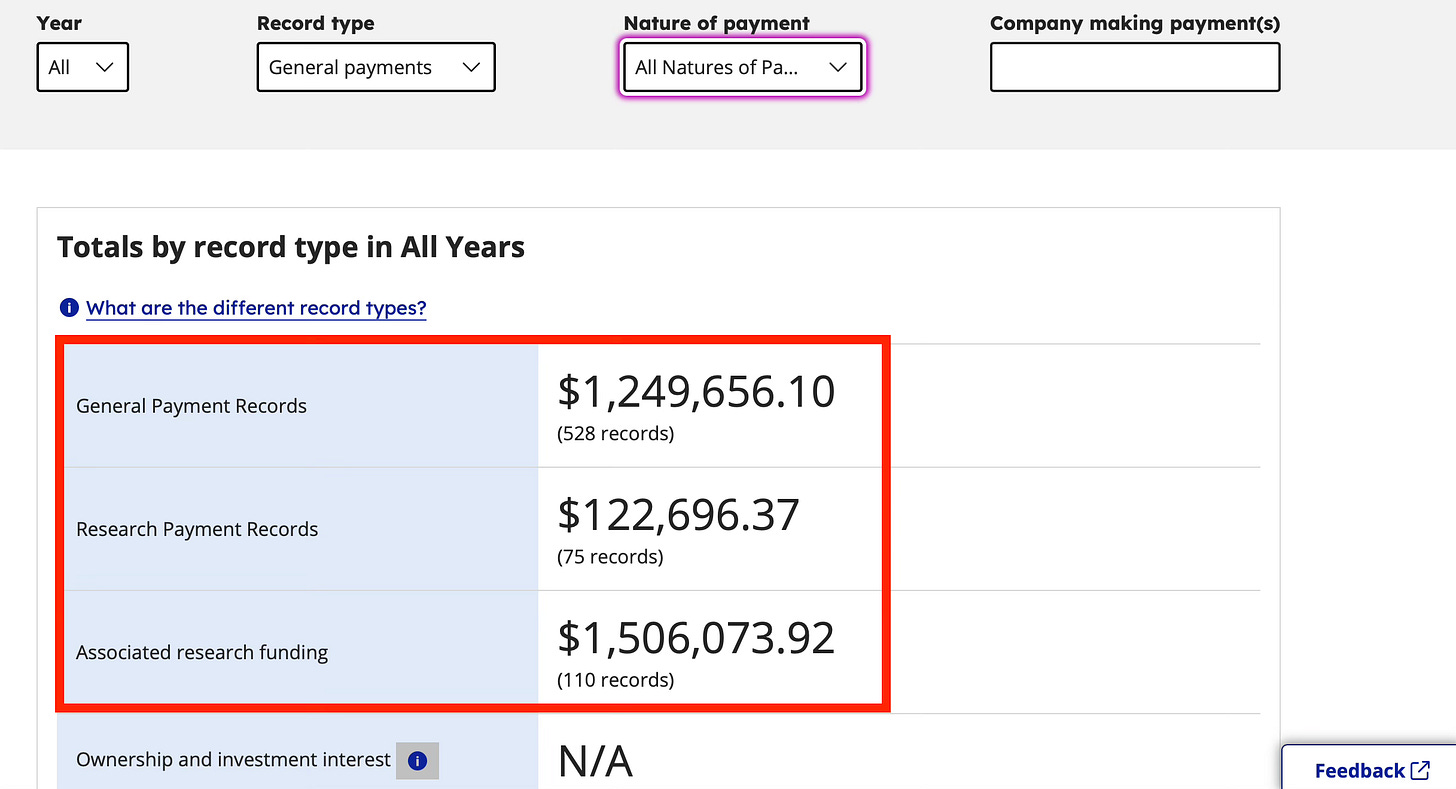
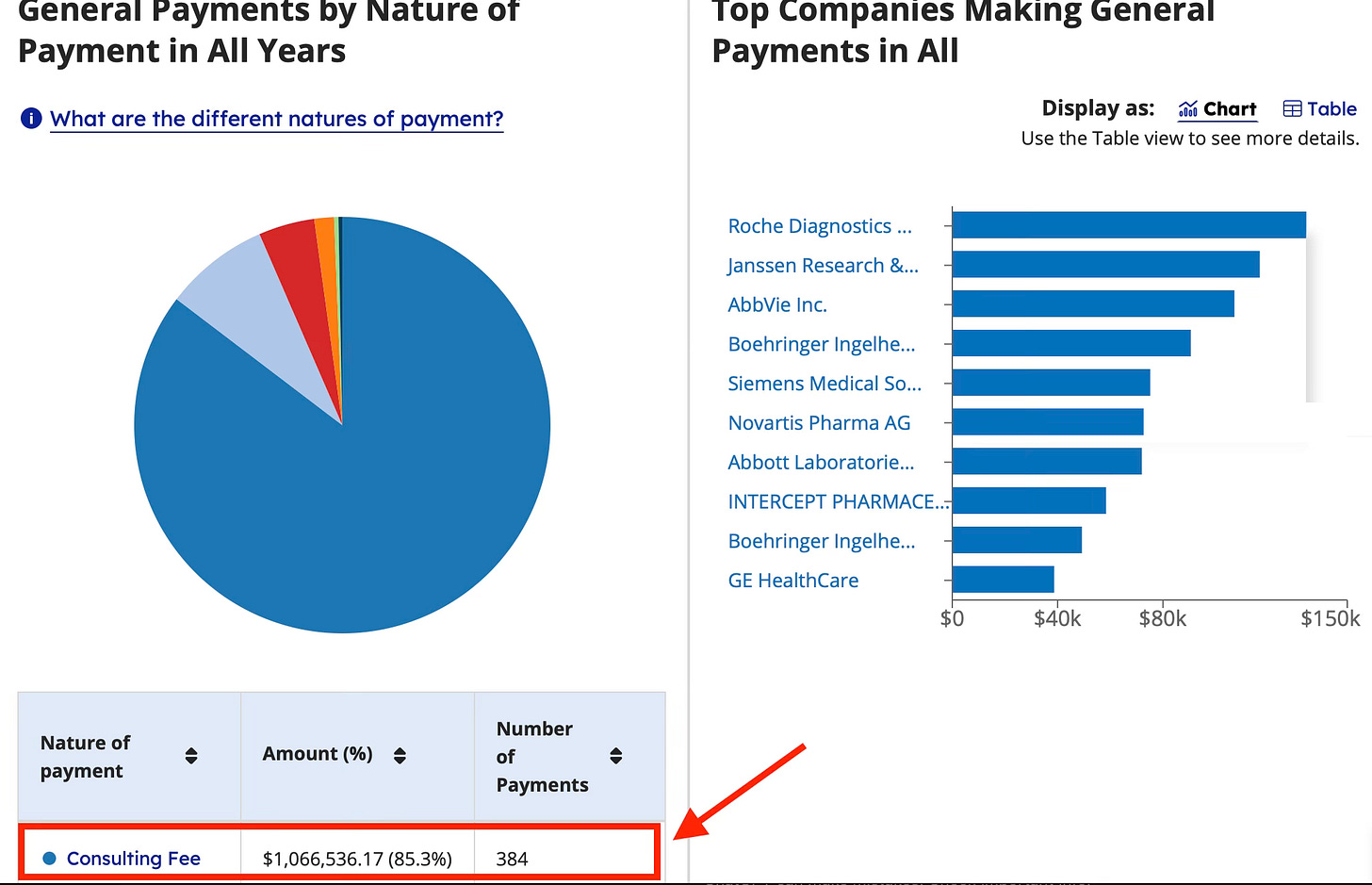
Let’s take a small detour and look at just one example from that table, Dr. James L. Januzzi Jr., a cardiologist at Massachusetts General Hospital and Harvard Medical School. His disclosure entry lists consulting roles with several diagnostics companies and participation in research or trial oversight roles connected to multiple pharmaceutical firms.
Here is Dr Januzzi on OpenPayments.gov.
Academic experts who conduct the clinical trials, publish the evidence, and advise professional societies on treatment recommendations often swim in broader research waters that are heavily funded by the pharmaceutical industry whose products those guidelines ultimately evaluate.
Aaaaaand the result was predictable in 2018. Industry funded the trials → the trials shaped the guidelines → the guidelines directed the therapeutical approach → the market followed … and lots of people got very rich.
So what do you think the new 2026 guidelines look like?
From The NY Times article Friday:
If the new guideline were widely followed, especially by younger people who could end up with a lifetime of lower LDL levels, there could be a big effect, cardiologists say.
The group recommends starting to test for levels of LDL cholesterol — the “bad” cholesterol — during childhood, at around age 10. Doing so could identify the one in 250 people with a genetic condition that leads to extremely high cholesterol levels. They would need to be immediately prescribed a cholesterol-lowering drug.
Earlier testing! And more testing!
Well OF COURSE that’s the answer!!! Hey, I bet the next guidelines will advise to start testing fetuses in utero! I mean, shit, let’s get a vaccine going in the pipeline. And we can start taking cholesterol boosters every other quarter with the covid shit shots - I mean, I think we could probably call them something more appropriate though. Cholesterol booster probably wouldn’t sell. Cholesterol dimmer kinda hits … 🤣
What else do the new guidelines say?
NYT continues…
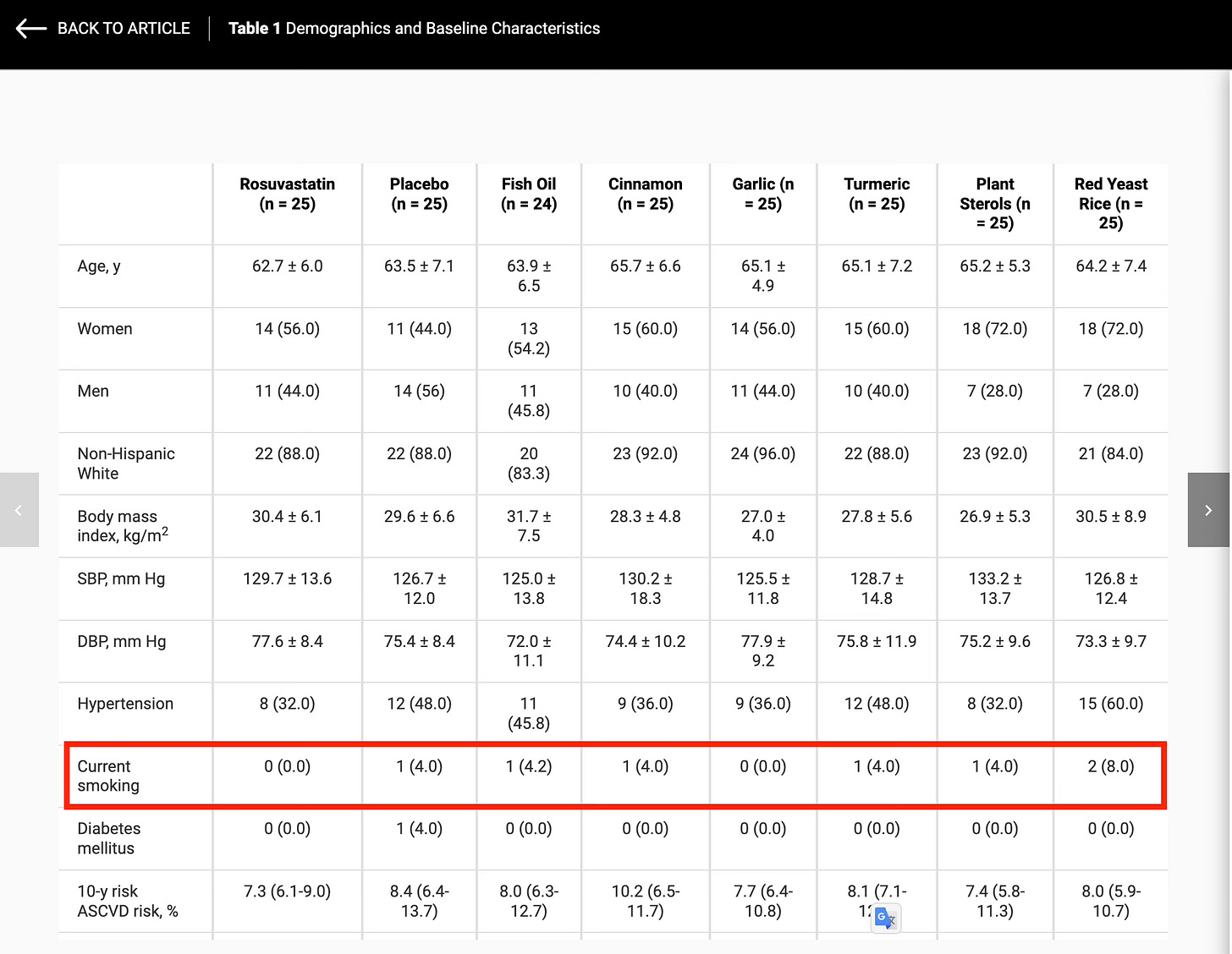
The guideline also says people should not rely on supplements to lower their LDL. A clinical trial, run by Dr. Nissen, tested six supplements. Five of them — fish oil, cinnamon, turmeric, plant sterols and red yeast rice — were no better than a placebo. The sixth — garlic in pill form — actually raised LDL levels.
Let’s just take a little look at the SPORT clinical trial, cited as evidence for discouraging the use of things other than pharmaceuticals.
Comparative Effects of Low-Dose Rosuvastatin, Placebo, and Dietary Supplements on Lipids and Inflammatory Biomarkers
Authors: Luke J. Laffin, Dennis Bruemmer, Michelle Garcia, Danielle M. Brennan, Ellen McErlean, Douglas S. Jacoby, Erin D. Michos, Paul M Ridker, Tracy Y. Wang, Karol E. Watson, Howard G. Hutchinson, and Steven E. Nissen
First of all, let’s have a round of applause for Gina Kolata of the New York Times and her journalistic prowess 👏. Excellent job parroting the narrative without doing any sort of digging that might resemble investigation.
👇Here’s what that would look like.
A total of 190 participants completed the study, which means a scant 25 people were in each cohort. Just in case you are unfamiliar with clinical trials, these numbers are quite small, making it highly sensitive to baseline imbalances. For example, while 3.5% of participants were current smokers, none were assigned to the Rosuvastatin arm. 33% were former smokers, but who cares. Apparently nobody, because that wasn’t even noted in the table.
With the scant 25 person sample size, even just a few unevenly distributed risk factors (like 1 or 2 smokers) could influence outcomes. Speaking of outcomes…we are, of course, in surrogate endpoint land, not actual clinical outcome land. I mean, with that stunning 28 day trial length, designed to detect large statin-like effects over a very short time period. Really, any possibility of slower metabolic changes from supplements being detected is statistically almost impossible with such small groups and short follow-up. This trial was set up to show some Real Science™. So there’s that.
Also this:
Patients taking 1 of the studied supplements before trial enrollment were allowed to participate if they discontinued for 4 weeks before enrollment.
Some supplements have longer physiological effects than 4 weeks, especially those that influence lipid metabolism.
For example, red yeast rice. It contains monacolin K, which is chemically identical to lovastatin, and the LDL-lowering effects can last at least several weeks after discontinuation. Or fish oil, a commonly used supplement, has omega-3s that incorporate into cell membranes. These effects can last months.
So if someone had been taking a supplement long-term, a 4 week washout might not fully eliminate its biological effects. But whatever, who cares.
Now let’s look at the disclosures, of which there are many …
Funding Support and Author Disclosures
This trial was funded by AstraZeneca Pharmaceuticals. Dr Laffin has been a consultant and/or served on steering committees for Medtronic, Lilly, Mineralys Therapeutics, AstraZeneca, and Crispr Therapeutics; has received research funding from AstraZeneca; and has ownership interest in LucidAct Health and Gordy Health. Dr Bruemmer has been a consultant and served on a steering committee for Esperion. Dr Jacoby has been a consultant and served on a steering committee for AstraZeneca. Dr Michos has been a consultant and/or served on steering committees for AstraZeneca, Bayer, Boehringer Ingelheim, Esperion, Novartis, Novo Nordisk, and Pfizer; and has received research funding from the American Heart Association and the National Institutes of Health. Dr Ridker has been a consultant and/or served on steering committees for Agepha, AstraZeneca, Civi Biopharm, Flame Inc, Health Outlook, IQVIA, Janssen Pharmaceuticals, Novartis, Novo Nordisk, Omeicos, and Sirnanomics; and has received research funding from Amarin, Kowa Pharmaceuticals, the National Heart, Lung, and Blood Institute, and Novartis. Dr Wang has been a consultant and/or served on steering committees for AstraZeneca and Novartis; and has received research funding from Abbott Laboratories, Artivion, Boston Scientific, Bristol Myers Squibb, and Chiesi. Dr Watson has been a consultant and/or served on steering committees for Amari, Amgen, Boehringer Ingelheim, Esperion, Novartis, and Lilly; and has been a speaker for Boehringer Ingelheim. Dr Hutchinson is an employee of AstraZeneca. Dr Nissen has received institutional research funding from AbbVie, AstraZeneca, Amgen, Bristol Myers Squibb, Cerenis, Eli Lilly, Esperion, Medtronic, MyoKardia, Novartis, Pfizer, The Medicines Company, Silence Therapeutics, Takeda, and Orexigen; and is an unpaid consultant for Respira, Alnylam, Beren Therapeutics, Prolaio, TenSixteen Bio, Altimmune, MetroBiotech, and Intercept. All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.
Well isn’t it so weird that the study funded by the company that makes a cholesterol drug, comparing that cholesterol drug to inexpensive supplements, where 75% of the authors were financially tied to companies that make cholesterol medications, notably the one funding the trial … found that the cholesterol drug is best.
Dr. Laffin all the way to the bank.
And I love the last sentence, ”All other authors have reported that they have no relationships relevant to the contents of this paper to disclose.” 👏🤗
All 3 other authors. 😂
Of course there were plenty of companies represented in the disclosure document that also make lucrative cholesterol drugs.
I encourage you to look into the work of Ben Goldacre. His book, Bad Pharma: How Drug Companies Mislead Doctors and Harm Patients, is about the pharmaceutical industry, its relationship with the medical profession, and the extent to which it controls academic research into its own products.
For example, in a “2007 journal article published in PLOS Medicine, researchers studied every published trial on statins, drugs prescribed to reduce cholesterol levels. In the 192 trials they looked at, industry-funded trials were 20 times more likely to produce results that favoured the drug”. (source)
Have a look at this paper, published in JAMA (2003):
Scope and Impact of Financial Conflicts of Interest in Biomedical Research
Conclusions: Financial relationships among industry, scientific investigators, and academic institutions are widespread. Conflicts of interest arising from these ties can influence biomedical research in important ways.
Check out this bullshit, published by The Lancet this year.
Assessment of adverse effects attributed to statin therapy in product labels: a meta-analysis of double-blind randomised controlled trials
I’ll summarize:
💁🏻♀️ Cherry picked data held by the CTT group that they won’t share with anyone ever, finds that statin adverse effects are all in your head and maybe drug companies should consider changing their labels so you don’t create a confirmation bias situation … you know, for all those people imagining side effects. 🫨
And the beat goes on.
Pharmaceutical companies just continue to market their drugs through thinly veiled propaganda, delivered by salesmen in white coats, heralded as experts and revered as The Science™.
And apparently nobody is ever going to notice this in any meaningful way. Certainly not the New York Times 😂.
















Remedy: Never go to doctor, never take a test nor pill. You will live worry-free, happy life. And when it's your time to go, you just go. Do not play their game. Being afraid kills you faster than any 'sickness'.
Hilarious take on the insanity of the pHARMacy propaganda. It was an alert for me that my next dr appointment is probably going to result in still another reason for them to call me a difficult patient. They've been fretting about my lack of statin participation for a while now. Good to have more ammunition! 💕